NADPH (β-Nicotinamide Adenine Dinucleotide Phosphate, Reduced Form), also known as Reduced Coenzyme II, is a key reduced redox coenzyme and high-efficiency electron/proton donor in organisms. As the reduced form of NADP⁺ (Oxidized Coenzyme II), it plays an irreplaceable role in biological metabolism, enzymatic catalysis, and antioxidant defense. As a high-value biochemical raw material, NADPH is widely used in high-end fields such as biomanufacturing, pharmaceutical intermediates, biological reagents, and medical research, and is a core supporting material for enterprises to achieve green catalysis and technological upgrading.
I. Core Basic Information (Chemical Grade Standard)
|
Item |
Specification Parameters |
Remarks |
|
Chinese Name |
β-Nicotinamide Adenine Dinucleotide Phosphate (Reduced Form), Reduced Coenzyme II, NADPH |
Unified industry designation, reduced form of NADP⁺, with strong reducibility, also known as reducing hydrogen |
|
English Name |
β-Nicotinamide Adenine Dinucleotide Phosphate (Reduced Form), NADPH |
Suitable for international procurement, foreign trade export and English technical documents |
|
CAS No. |
53-57-6 (Free Acid Exclusive CAS No.) 2646-71-1 (β-NADPH, Coenzyme II reduced tetrasodium salt) |
Different from monosodium salt (100929-71-3), can be accurately traced |
|
Molecular Formula/Molecular Weight |
C₂₁H₂₉N₇O₁₇P₃ / 744.42 (Free Acid Standard) C21H26N7Na4O17P3(tetrasodium salt) |
Stable structure, excellent reducibility, suitable for enzymatic catalysis and scientific research scenarios |
|
Appearance |
White to pale yellow crystalline powder, odorless, with strong hygroscopicity |
When purity ≥98%, the appearance is uniform without impurities and caking, and the dry product has better stability |
|
Solubility |
Easily soluble in water and methanol, slightly soluble in ethanol, insoluble in ether and ethyl acetate |
The aqueous solution has the best stability at pH 6.0-7.5, with no obvious degradation within 24 hours |
|
Purity Standard |
≥98% (HPLC detection) |
Complies with dual standards of biochemical reagents and pharmaceutical grade, controllable impurities, suitable for high-precision scenarios |
|
Stability |
Sealed, protected from light and dry storage at 2-8℃ (24 months); long-term storage at -20℃ (36 months) |
Better stability when stored in cold place, avoid high temperature, strong light and oxidants to prevent oxidative degradation |
|
Impurity Control |
Heavy metals ≤0.5ppm, nucleic acid/protein impurities ≤0.1%, NADP⁺ impurities ≤1.0% |
Suitable for high-precision enzymatic catalysis, pharmaceutical intermediates, scientific research and testing and other high-end scenarios |
II.Structure and Core Characteristics
1.Chemical Structure
NADPH is composed of nicotinamide mononucleotide (NMN) and 5′-adenosine monophosphate connected by a pyrophosphate bond. Compared with NADH, it has an additional phosphate group on the adenine ribose. The nicotinamide ring is the core active center, and the hydrogen atoms on the ring endow NADPH with strong reducibility, which can reversibly transfer electrons and protons to complete the mutual conversion between NADPH and NADP⁺, providing a structural basis for its role as an electron donor in enzymatic catalysis and antioxidant reactions.
2.Core Chemical Characteristics
- Strong Reducibility: As a high-efficiency electron/proton donor, it can quickly transfer electrons and protons to drive reduction reactions. It is a key reducing agent in the biological synthesis process, and can adapt to various reduction reaction scenarios when cooperating with oxidoreductases.
- Good Solubility: Easily soluble in water and common buffer solutions, can quickly dissolve to form a stable system, suitable for scientific research experiments, small-scale catalysis, pharmaceutical formulations and other scenarios, without the need for additional cosolvents.
- Good Structural Stability: Not easy to oxidize and degrade under suitable storage conditions, maintains active stability in the pH range of 5.0-8.0, can adapt to most enzymatic catalysis and scientific research experimental systems, and cold storage can extend the stability period.
- Good Compatibility: Can be compatible with various enzyme preparations, buffer solutions and pharmaceutical intermediates without antagonistic reactions, can cooperate with a variety of catalysts to help build an efficient cycle catalytic system, and does not interfere with the stability of the reaction system.
- Environmental Protection: As a biological coenzyme, it can achieve cycle regeneration in combination with enzymes, no need for additional supplementation, reduce the generation of chemical waste, conform to the concept of green production and scientific research environmental protection, and reduce the use cost.
III. Mechanism of Action, Core Effects and Application Fields
1.Core Mechanism of Action
As a reduced coenzyme, the core mechanism of NADPH is to reversibly accept and transfer electrons/protons through the nicotinamide ring. It is oxidized to NADP⁺ under enzymatic catalysis, and NADP⁺ can be reduced to NADPH under the action of dehydrogenases (such as glucose dehydrogenase, 6-phosphoglucose dehydrogenase) to complete the “oxidation-reduction” cycle regeneration. It can realize hydrogen transfer with NAD⁺ through transhydrogenase, indirectly participate in the respiratory chain process, continuously provide electron donors for reduction reactions, and greatly reduce production and scientific research consumption without additional supplementation of coenzymes.
2.Core Effects
- Catalytic Synergy: Cooperates with various oxidoreductases to improve the rate and selectivity of catalytic reactions, reduce by-products, increase the yield of target products, and adapt to the fine synthesis of chiral drugs and high-value-added chemicals.
- Antioxidant Protection: Can reduce oxidized glutathione (GSSG) to reduced glutathione (GSH), scavenge reactive oxygen species (ROS), protect enzyme activity and target products from oxidative degradation, and support the construction of antioxidant systems.
- Metabolic Regulation: Provides reducing equivalents for the synthesis of fatty acids, cholesterol, nucleotides and other substances, adapts to scientific research scenarios such as biological metabolism simulation, cell culture, and photosynthesis-related research, and supports small-scale synthesis needs.
- Cycle Regeneration: Combines with enzyme systems to achieve closed-loop catalysis, which can be recycled by itself, reduce coenzyme consumption, reduce production costs and waste of scientific research reagents, and conform to the development trend of green chemical industry and scientific research.
3.Main Application Fields
With excellent reducibility and biocompatibility, NADPH is widely used in fields such as enzymatic catalysis, medicine, and scientific research, especially suitable for enzyme cycle regeneration catalysis and scientific research detection scenarios. Specific reaction examples (including reaction formulas) are as follows:
- Example 1: NADPH + Carbonyl Reductase Combination — Catalyzes the reduction of ketone compounds (such as acetophenone) to chiral alcohols (phenylethanol). NADPH provides electron donors, and after being oxidized to NADP⁺, it is reduced to NADPH under the action of glucose dehydrogenase (GDH) and glucose to achieve cycle regeneration.
Reaction Equations (Two-Step Cycle):Total Reaction:(NADPH is recycled and not consumed)
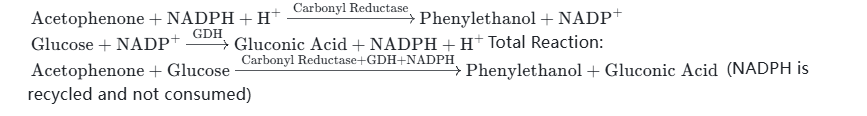
- Example 2: NADPH + Glutathione Reductase (GR) Combination — Reduces oxidized glutathione (GSSG) to reduced glutathione (GSH), which is used in the construction of pharmaceutical antioxidant systems and scientific research on antioxidant. After NADPH is oxidized to NADP⁺, it is reduced and regenerated through the action of malate dehydrogenase (MDH) and oxaloacetic acid.
Reaction Equations (Two-Step Cycle):
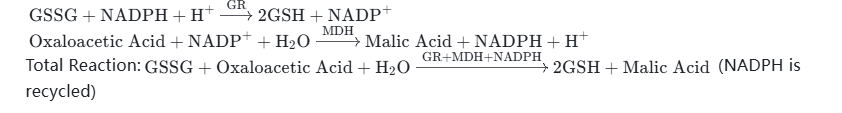
Total Reaction:(NADPH is recycled) - Example 3: NADPH + Nitrate Reductase Combination — Catalyzes the reduction of nitrate (NO₃⁻) to nitrite (NO₂⁻), which is used in the synthesis of food preservatives and scientific research related to environmental detection. After NADPH is oxidized to NADP⁺, it is reduced and regenerated under the action of isopropanol dehydrogenase (ADH) and isopropanol.
Reaction Equations (Two-Step Cycle):
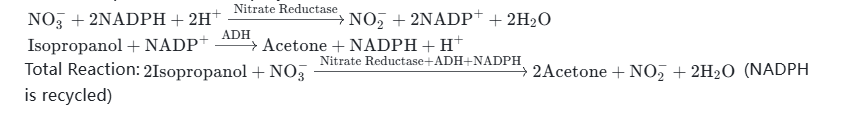
Total Reaction:(NADPH is recycled) - Example 4: NADPH + 6-Phosphoglucose Dehydrogenase (G6PDH) Combination — Catalyzes the oxidation of 6-phosphoglucose to 6-phosphogluconolactone, provides reducing equivalents for the synthesis of steroid drugs, and is also suitable for photosynthesis-related scientific research. After NADPH is oxidized to NADP⁺, it can be reduced and regenerated through glucose and GDH enzyme to form a closed-loop catalytic system.
Reaction Equations (Two-Step Cycle):
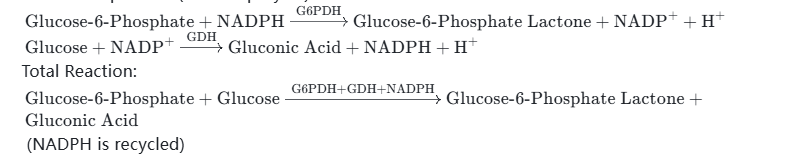
Total Reaction:(NADPH is recycled) - Example 5: NADPH + Lactate Dehydrogenase (LDH) Combination — Catalyzes the reduction of pyruvate to lactic acid (food/pharmaceutical raw material), which is suitable for metabolic research and small-scale synthesis scenarios. After NADPH is oxidized to NADP⁺, it can be reduced and regenerated under the reverse catalysis of pyruvate and LDH to achieve cycle catalysis and reduce production costs.
Reaction Equations (Two-Step Cycle):

Total Reaction:(NADPH is recycled and not consumed)
IV. Product Advantages (Factory Direct Supply)
- Controllable and Traceable Purity: Adopting advanced purification technology, each batch of products passes full-item detection such as HPLC, Nuclear Magnetic Resonance (NMR), and heavy metals. The purity is stably ≥98%, and 99.9% high purity can be customized. NADP⁺ impurities are strictly controlled ≤1.0%, ensuring excellent reducibility and catalytic activity, suitable for scientific research and high-end industrial scenarios.
- Customized Services: Supports customization of free acid form and purity grading, provides various packaging specifications (g/kg level), adapts to different needs such as scientific research experiments and small-scale production, and can provide technical support to optimize usage plans and reaction systems.
- Stable Supply Chain: Own production base, passed ISO9001, FDA registration and other international certifications, spot supply, shortened delivery cycle, reduced procurement costs, ensured stable supply of high-end raw materials, and supported the continuous progress of scientific research projects and production.
- Compliance and Technical Support: Complete qualifications, in line with EU, US and other international standards, can provide a full set of test reports and technical documents; professional team provides full-process technical guidance, including enzymatic reaction optimization, formula adaptation, etc., to help product upgrading and scientific research project implementation.
- Stable Quality: Optimized production and purification processes, effectively improved the stability of NADPH, reduced oxidative loss during storage and use, ensured the consistency of product performance, and adapted to various scientific research and catalytic scenarios.
V. Storage and Usage Instructions
1. Storage Conditions
- Powder: Store sealed, protected from light and dry at 2-8℃, avoid repeated freezing and thawing, and isolate from oxidants; long-term storage at -20℃ can extend the shelf life to 36 months, and cold storage can better maintain its reducibility.
- Solution: Prepare and use immediately; if temporary storage is needed, it can be stored at 4℃ for 7 days, and stored frozen in aliquots at -20℃ for 1 month, protected from light and isolated from air to prevent oxidative degradation affecting the use effect.
2.Usage Notes
- Dissolution: Dissolve with sterile water, normal saline or buffer solution (pH 6.0-7.5), the addition temperature in the formula ≤45℃, avoid high temperature damaging the structure and reducing reducibility.
- Compatibility: Avoid mixing with strong oxidants, strong acids and strong alkalis to prevent oxidative degradation; when compatible with enzyme preparations, optimize pH and temperature conditions to improve catalytic efficiency.
- Safety: Wear protective equipment during operation; prohibited for non-industrial, non-scientific research, non-medical research purposes, not for direct consumption, and strictly follow the operation specifications of chemical raw materials.
- Operation: Avoid prolonged exposure to air and strong light during use, and seal the remaining products in time to reduce oxidative loss and ensure the subsequent use effect.
VI. Qualification and Testing
Our NADPH products have passed strict testing, and we can provide a full set of qualification documents such as HPLC purity report, heavy metal testing, microbial testing, NADP⁺ impurity testing. The products comply with industry standards in multiple fields such as biological reagents, medicine, cosmetics, and medical research, with controllable and traceable quality, ensuring the compliance and stability of high-end application scenarios, and adapting to high-end industrial and scientific research application needs such as enzymatic catalysis and scientific research testing.

